PsbA-NC PCR #6 8/13/19
Testing Diluting primers
Procedure
PsbA-NC is a plastid primer - Primer info
Samples E1 E2 E3 E4 E5 E6 E7 were used in this PCR (polimerase chain reaction) reaction
Two master mixes were made A Master Mix of water, 2x mytaq mix, 1,788F Primer (concentration: 1 and 5 micromolar), 2,218R Primer (concentration: 1 and 5 micromolar)
| Component | Volume for 7 reactions(+5% error) |
|---|---|
| Water | 66.15 micoleters |
| mytaq mix | 91.9 micoleters |
| 1,788F Primer (concentration: 1 micromolar) | 7.35 micoleters |
| 2,218R Primer (concentration: 1 micromolar) | 7.35 micoleters |
| BAS (Borine Serum Albumin) | 1 microliter |
| Component | Volume for 7 reactions(+5% error) |
|---|---|
| Water | 66.15 micoleters |
| mytaq mix | 91.9 micoleters |
| 1,788F Primer (concentration: 5 micromolar) | 7.35 micoleters |
| 2,218R Primer (concentration: 5 micromolar) | 7.35 micoleters |
| BAS (Borine Serum Albumin) | 1 microliter |
24 micoleters of this master mix was added to 7 tubes For the 2 negative controls of 25 micoleters water no positive control was available
1 microleter of the DNA samples was added to the coresponding tube
The samples were then placed in the thermocycler set to
| Step | Temperature | Duration | cycles |
|---|---|---|---|
| 1 | 94°C | 2:00 | 1 |
| 2 | 64 | 1:30 | 1 |
| 3 | 72°C | 2:00 | 1 |
| 4 | 94°C | 0:45 | 36 |
| 5 | 64 | 1:00 | 36 |
| 6 | 72°C | 1:30 | 36 |
| 7 | 72°C | 2:00 | 1 |
| 8 | 4°C | hold | - |
these amplified DNA samples were then tested by making a gel
to create the Gel the following steps were taken
1.5g agarose and 75ml 1X TAE buffer were mixed to make a 2% agarose 75ml gel mix the mix was microwaved and swirled until there were no flakes then 1 microliters Gelgreen was added
the mix was left to cool for 5 minutes then poured into the gel mold the gel was left to harden for 30 minutes
once hardened enough Used TAE buffer was poured into the gel box to cover the mold
5 microliter aliquots of DNA samples E1 E2 E3 E4 E5 E6 E7 and the two controls were made 1 microliter of 5x loading dye was added to each
First 3 microliters of the HyperLadder 100bp was added to the first well of the mold then the DNA samples were each added to the wells in the following order
| Ladder | E1 | E2 | E3 | Negative Control | E4 | E5 | E6 | E7 |
| Ladder | E1 | E2 | E3 | Negative Control | E4 | E5 | E6 | E7 |
The voltage was set to 100V and run for 60 minutes
This is the resulting Gel
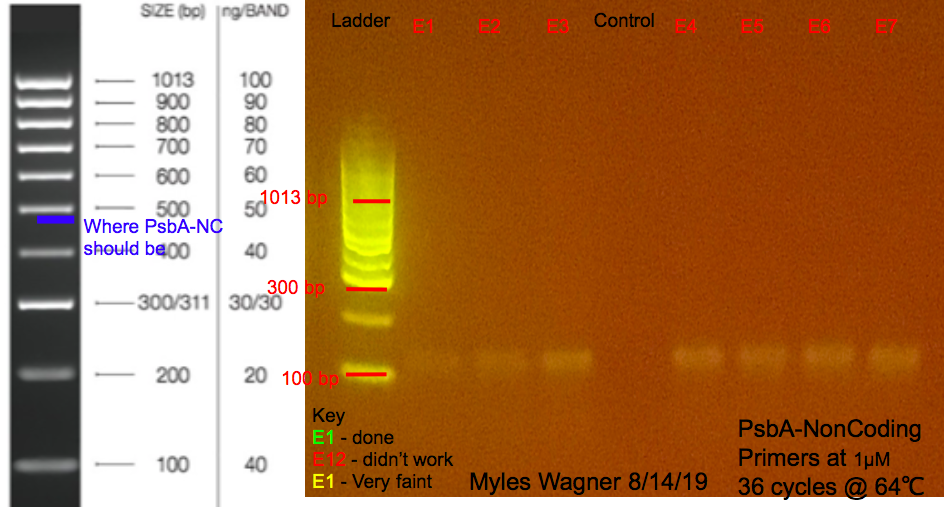
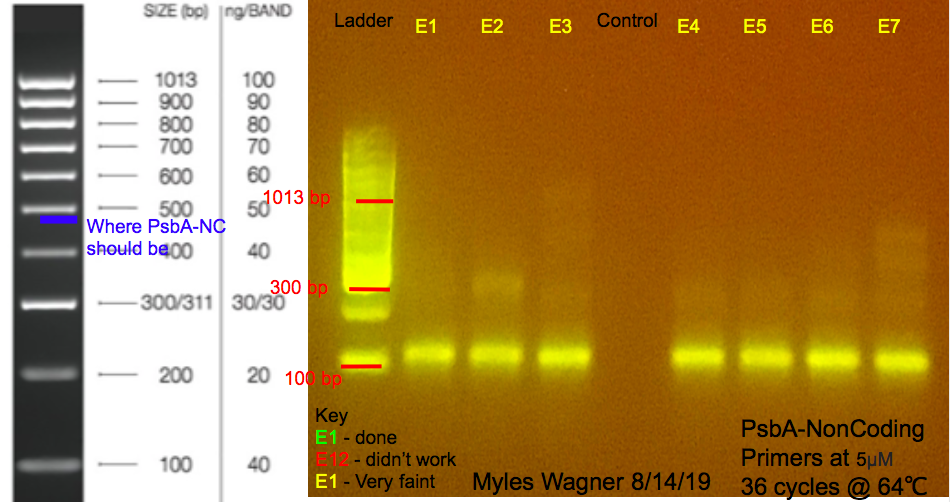
Notes
this was to try to get rid of the secound band on all the previous PCRs